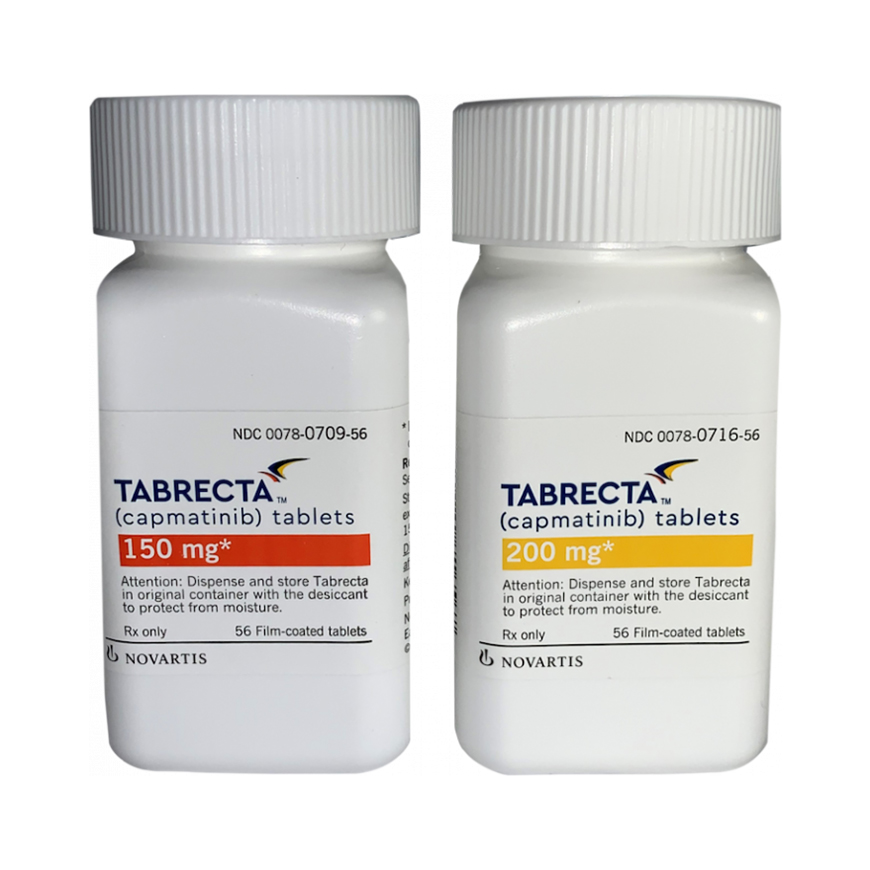
Best Capmatinib tablet 200 mg Tabrecta price in india at yourmedikart Manufactured by Novartis |.
Tabrecta 200 Mg Tablets
Novartis
Generic Name : Capmatinib Tablets
Packing : 56 Film Coated Tablets

What is the use of Tabrecta ?
- Tabrecta medicine treat lung cancer (NSCLC) that spread of the body or cannot removed by surgery (metastatic). The tumors have abnormal mesenchymal epithelial progression (MET) genes.
- Learn more about the effective Tabrecta 200 mg treatment option and its uses. Explore the specific Tabrecta 200 mg Tablet for reliable results. Discover the benefits of Capmatinib and check the availability of Capmatinib Tablets.
- Compare the affordable Tabrecta Price and get details about Tabrecta from trusted sources. Learn about the Tabrecta Manufacturer and explore the competitive Capmatinib Cost.
- For dosage information, refer to the detailed Tabrecta Dosing, and access official prescribing details with the Tabrecta PI.
What is Tabrecta?
Tabrecta is a medicine prescribed by doctor use to treat adults with a type of lung cancer called non-small cell lung cancer (NSCLC):
- Has spread to different parts of the body or can’t take out by a surgery (metastatic), and
- Whose tumors have an abnormal mesenchymal epithelial transition (MET) gene.
Buy Tabrecta Online at yourmedikart.store Tabrecta cost is low in India.
Indications
- The drug show in FDA-clear tests for treating patients with metastatic non-small cell lung cancer (NSCLC) who have a tumor with a mutation that causes mesenchymal-epithelial metastasis (MET) different exon 14 cross.
- This sign approved under accelerated approval dependent on overall response rate and length of reaction. Proceeded with approval for this sign might dependent upon verification and description of clinical advantage in confirmatory trial(s).
Buy Tabrecta Online at yourmedikart.store. Tabrecta cost is low in India.
Before taking Tabrecta
Before taking Tabrecta, tell your healthcare provider about all of your medical conditions, including if you:
- Have or have had lung or breathing problems other than your lung cancer.
- Have or have had liver problems.
- Are pregnant or plan to become pregnant. Tabrecta can harm your unborn baby.
- Females who are able to become pregnant:
- Your healthcare provider should do a pregnancy test before you start treatment.
- You should use effective birth control during treatment and for 1 week after your last dose. Talk to your healthcare provider about birth control choices that might be right for you during this time.
- Tell your healthcare provider right away if you become pregnant or think you may be pregnant during treatment.
- Males who have female partners who can become pregnant:
- You should use effective birth control during treatment and for 1 week after your last dose.
- Are breastfeeding or plan to breastfeed. It is not known if Tabrecta passes into your breast milk. Do not breastfeed during treatment and for 1 week after your last dose.
- Tell your healthcare provider about all the medicines you take or start taking, including prescription and over-the-counter medicines, vitamins, and herbal supplements.
How should I take Tabrecta?
- Take Tabrecta 200 mg exactly as your healthcare provider tells you.
- Take Tabrecta tablets 2 times a day with or without food.
- Swallow the tablets whole. Do not break, chew, or crush the tablets.
- Your healthcare provider may change your dose, temporarily stop, or permanently stop treatment if you have certain side effects.
- Do not change your dose or stop treatment unless your healthcare provider tells you to.
- If you miss or vomit a dose, do not make up the dose. Take your next dose at your regular scheduled time.
Dosage
Patient Selection
Select patients for treatment dependent on the presence of a mutation that prompts MET exon 14 avoiding in tumor examples. Data capmatinib 200 mg tablet on FDA-approved tests is accessible at Companion Diagnostics.
Suggested Dosage
- The suggested dosage of TABRECTA is 400 mg orally twice every day with\ without food.
- Secondly Swallow TABRECTA tablets. Try not to break, crush or chew the tablets.
- Thirdly If a patient misses or vomits a dose, capmatinib 200 mg tablet instruct the patient not to make up the dose, but rather to take the following dose at its scheduled time.
Tabrecta Side Effects
Most common Tabrecta side effects:
- Loss of appetite
- Weakness and tiredness
- Swelling of your hands or feet
- Changes in certain blood tests
- Vomiting
These are the most common side effects of Tabrecta Cena. Capmatinib tablet 200 mg Tabrecta price in india at yourmedikart
What are the possible side effects of Tabrecta?
Tabrecta may cause serious side effects, including:
- lung or breathing problems.
- Tabrecta may cause inflammation of the lungs that can cause death. Tell your healthcare provider right away if you develop any new or worsening symptoms, including:
- Cough
- Fever
- Trouble breathing or shortness of breath
Your healthcare provider may temporarily stop or permanently stop Tabrecta if you develop lung or breathing problems during treatment.
- Tabrecta may cause inflammation of the lungs that can cause death. Tell your healthcare provider right away if you develop any new or worsening symptoms, including:
- liver problems.
- Tabrecta may cause abnormal liver blood test results. Your healthcare provider will do blood tests to check your liver function before you start, and during treatment. Tell your healthcare provider right away if you develop any signs and symptoms of liver problems, including:
- Your skin or the white part of your eyes turns yellow (jaundice)
- Loss of appetite for several days or longer
- Dark or “tea-colored” urine
- Nausea and vomiting
- Light-colored stools (bowel movements)
- Confusion
- Pain, aching, or tenderness on the right side of your stomach-area (abdomen)
- Tiredness
- Weakness
- Swelling in your stomach-area
Your healthcare provider may change your dose, temporarily stop, or permanently stop treatment if you develop liver problems.
- Tabrecta may cause abnormal liver blood test results. Your healthcare provider will do blood tests to check your liver function before you start, and during treatment. Tell your healthcare provider right away if you develop any signs and symptoms of liver problems, including:
-
Risk of sensitivity to sunlight (photosensitivity)
- The most common side effects include:
- Swelling of your hands or feet
- Nausea
- Tiredness and weakness
- Vomiting
- Loss of appetite
- Changes in certain blood tests
- The most common side effects include:
Drug Interactions
Effect of Other Drugs on Tabrecta
Strong CYP3A Inhibitor
- Co-administration of TABRECTA and a strong CYP3A inhibitor increases the exposure of capmatinib, which may increase the incidence and severity of TABRECTA adverse reactions [see Clinical Pharmacology]. Closely monitor patients for adverse reactions during the co-administration of TABRECTA with strong CYP3A inhibitors.
- Strong and moderate CYP3A inducers TABRECTA co-administration with strong CYP3A inducers reduced capmatinib exposure. Co-administration of TABRECTA and moderate CYP3A inducers may also reduce capmatinib exposure. Reducing the exposure to capmatinib may reduce the anti-tumor activity of TABRECTA. Avoid co-administration of TABRECTA with strong and moderate CYP3A inducers.
Effect of Tabrecta on Other Drugs
- Co-administration of the CYP1A2 substrate TABRECTA increases the exposure of CYP1A2 substrates. Most Importantly which may increase the adverse reactions of these substrates.
- Similarly If co-administration between TABRECTA and CYP1A2 substrate is inevitable, and small changes in concentration can cause serious adverse reactions, reduce the dose of CYP1A2 substrate according to the approved prescribing information.
- TABRECTA increases the exposure of P-gp and BCRP substrates, capmatinib 200 mg tablet price which may increase the adverse reactions of these substrates [see Clinical Pharmacology].
More Detail
- Because If co-administration between TABRECTA and P-gp or BCRP substrate is unavoidable, and small changes in concentration may cause serious adverse reactions, please reduce the dose of P-gp or BCRP substrate according to the approved prescribing information. Exposure of MATE1 and MATE2K substrates can increase the adverse reactions of these substrates.
- But if the co-administration of TABRECTA and MATE1 or MATE2K substrate is unavoidable. And small changes in concentration can cause serious adverse reactions, please reduce the dose of MATE1 or MATE2K substrate according to the approved prescribing information.
Warnings and Precautions
Precautions
- Interstitial Lung Disease (IP)/Pneumonia Pneumonia/IP can fatal and occurs in patients treated with TABRECTA. GEOMETRY mono-1 developed ILD/pneumonia in 4.5% of patients treated with this drug, 1.8% of patients developed grade 3 ILD/pneumonia and one patient died (0.3%). Similarly capmatinib 200 mg tablet price The median time to onset of grade 3 or higher ILD/pneumonia was 1.4 months (range: 0.2 months to 1.2 years).
- Look for signs of new or Worsening lung disease (such as Shortness of breath, cough, fever) indicating ILD/Pneumonia. Meanwhile Stop this drug Immediately capmatinib 200 mg tablet for Patients with Suspected ILD/pneumonia. Permanent exclusion unless other causes of PID/pneumonia have identified
Hepatotoxicity
Hepatotoxicity occurs in patients treated with TABRECTA [see Adverse Reactions].3 patients (0.9%) were discontinued from this treatment. The average height of grade 3 or higher ALT/AST was 1.4 months (maximum: 0.5 to 4.1 months). Monitor liver function before starting, every 2 weeks for the first 3 months of treatment, then monthly for patients with high dose transaminases or on treatment or frequent bilirubin. Discontinue, reduce dose, or discontinue However, TABRECTA permanently depending on severity of reaction [see Dosage and Administration].
Photosensitivity Hazards
- According to the results of animal studies, TABRECTA has a potential risk of photosensitivity [see Non-Clinical Toxicology]. In GEOMETRY mono-1, patients are advised to take preventive measures to prevent UV exposure, such as wearing sunscreen or protective clothing during So treatment. Because it is recommended that patients limit direct UV exposure during TABRECTA treatment.
You Will Get Capmatinib tablet 200 mg Tabrecta price in india at yourmedikart
Drug Interactions
Embryo-fetal toxicity
- According to the results of animal studies and its mechanism of action, Tabrecta cost Capmatinib may cause fetal harm when administer to pregnant women. According to the area under the curve (AUC) clinical dose of 400 mg twice a day, oral administration of Capmatinib to pregnant rats and rabbits during the organ Formation period caused malformations, and the exposure was lower than so the human exposure. Inform pregnant women of the potential risks to the fetus.
- Because It is Recommended that women of Childbearing age take the correct vaccine at the same time and within one week of the last dose. Men whose partners are able to conceive are encouraged to take the appropriate vaccine while taking this medication and within a week of the last dose. Manufactured by Novartis Capmatinib tablet 200 mg Tabrecta price in india at yourmedikart .
Clinical trials on Tabrecta
- Firstly The FDA Approval of Capmatinib is base on the GEOMETRY mono-1 Phase II Multicenter, Open-label, Non-Randomized, Multi-cohort study.
- However, The study Evaluated a total of 97 adult patients, including 69. Previously Treated patients and 28 treatment-naïve patients after central Confirmation of the METex14 skipping. Enroll patients were referred to group 4 or 5b and received 400 mg twice daily until Infectious or non-infectious.
- But The study overall response rate (ORR) and Duration of response (DOR) as Efficacy Outcomes.
- The confirmed ORR was 41% and 68% among previously treated and Treatment-naïve patients, respectively.
- Median DOR was find 9.7 months in previously treat patients (28 Responders) and 12.6 months in treatment naïve patients (19 Responders). The most common side effects Observed in patients during the trial were nausea, tabrecta price in india Vomiting, fatigue, Shortness of breath, Decreased Appetite, and Peripheral edema.
Tabrecta Approvals
- Firstly Novartis submit the New Drug Application (NDA) for the use of Capmatinib to the FDA in December 2019. So, The MET Inhibitor Receive priority review from the FDA in February 2020.
- Secondly The FDA Approved the therapy under the Provisions of the Accelerated Approval Regulations base on overall response rate and Duration of response, in May 2020.
- It also Approved Foundation One CD for use as a Companion Diagnosis to aid in the Identification of Mutations that Contribute to METex14 Skipping in tumor tissue.
- For Instance, The Regulator previously granted Innovative therapy and Orphan drug Designations to Novartis for Capmatinib.
- In June 2020, Tabrecta Price received Approval from the Ministry of Health, Labor and Welfare of Japan (MHLW) for use in the treatment of advanced or Recurrent Unrespectable NSCLC with a positive METex14 Mutation.
- However, Capmatinib tablet 200 mg Tabrecta price in india at Yourmedikart Manufacture by Novartis
How should I store Tabrecta?
Explore Tabrecta price and Tabrecta cost, along with capmatinib price in India. Learn about Tabrecta 200 mg, capmatinib price, and Tabrecta 200 mg price. Find out about Tabrecta coupon, Tabrecta interactions, and Tabrecta dosing. Check information on Tabrecta Novartis, capmatinib dose, and capmatinib cost. Get details on Tabrecta PI, Tabrecta dosage, and capmatinib brand name. Learn about capmatinib side effects and the Tabrecta manufacturer for comprehensive knowledge.
- Firstly Store Tabrecta at room temperature between 68°F to 77°F (20°C to 25°C).
- Store tablets in the original package with the drying agent (desiccant) cartridge.
- Protect the tablets from moisture.
- Throw away (discard) any unused Tabrecta you have left after 6 weeks of first opening the bottle.
Keepall medicines out of the reach of children and pets.
FAQ
What is the use of Tabrecta ?
Tabrecta is doctor Prescribe Medication treat adults with a kind of lung cancer called non-small cell lung cancer (NSCLC) that: has spread to different parts of the body or can't take out by a surgery (Metastatic) whose tumors have a abnormal mesenchymal epithelial progress (MET) gene.
Is Tabrecta a pill?
What is Tabrecta? Tabrecta is a doctor prescribed medication treat adults with a kind of lung cancer called non-small cell lung cancer that: has spread to different parts of the body or can't take out by a medical procedure (metastatic)
Is Capmatinib Chemotherapy?
Capmatinib has appear to specifically block the effects of the MET gene and consequently may help in monitoring the disease, preventing cancer cells from developing. Docetaxel is a standard chemotherapy medication generally treat your type of lung cancer.
When was Tabrecta FDA approved?
On May 6, 2020, the Food and Drug Administration allowed accelerated approval to capmatinib (TABRECTA, Novartis) for adult patients with metastatic non-small cell lung cancer (NSCLC) whose tumors have a mutation that prompts mesenchymal-epithelial transition (MET) exon 14 skipping as recognized by a FDA-approved test.
What does Tabrecta do?
TABRECTA (capmatinib) tablets is recommended medication treat adults with a kind of lung cancer called non-small cell lung cancer (NSCLC) that: has spread to different parts of the body or can't remove by a medical procedure (metastatic), and. whose tumors have an Abnormal Mesenchymal-epithelial transition (MET) gene.